BOMBSHELL REPORT - Startling Spike in Singapore's Cancer Medication Sales in 2023!
Another Aussie17 Exclusive!
Singapore serves as an effective control group for analyzing mRNA vaccine-related side effects and fatalities.
In 2020, at the onset of COVID-19, Singapore registered only 29 deaths for the entire year and approximately 60,000 infections, attributable to stringent lockdowns and contact tracing. The Case Fatality Ratio (CFR) was less than 0.05%, in contrast with the World Health Organization's false and exaggerated rate of 3.4%. I have proposed some hypotheses here regarding their notably low CFR.
Leveraging digital technology, Singapore's Foreign Minister Vivian Balakrishnan has referred to it as "perhaps the most successful contact-tracing program in the world" in 2021.
The HART foundation also identified Singapore as a control group in the article below.
Singapore is one of the most boosted countries per 100 people in the world as of July 2022.
Japan and Chile eventually surpassed them, but in terms of speed, Japan was approximately a few months behind (Singapore began vaccinations on December 20, 2020, and Japan started middle of Feb 2021).
And although Chile had a very high vaccination rate, less than 25% of its population received took the DNA-contaminated mRNA (Japan and Singapore had more than 95% receive mRNA vaccines, particularly Pfizer's).
Even with the low number of COVID-19 cases and a minimal COVID fatalities in 2020, Singapore now holds the third position globally for quarterly excess mortality.
Today, I'd like to present to you data on cancer drug sales in Singapore, which will provide insights into future excess deaths trends. I've often mentioned that cancer is a lagging indicator—people don't typically die immediately after getting cancer, but a rising trend in cancer cases can be predictive of increased excess mortality in the future.
Before delving into the sales data, it's essential to understand the typical sales trajectory for pharmaceuticals. The cycle usually starts with a rapid increase in sales, illustrated by an S-Curve, before reaching a steady state. After a drug's patent expires, its sales often start to dwindle as generic versions become available. Rapid growth in the sales of some drugs doesn't necessarily indicate a higher cancer diagnosis rate; it could signify a new medication capturing market share from established drugs. Conversely, a decline in sales doesn't always mean a decrease in cancer prevalence.
However, noteworthy consideration arises when a drug deviates from its expected pattern—particularly an older medication with consistently decreasing sales that suddenly experiences a surge.
Such an anomaly warrants further investigation.
Additionally, the data I'm presenting is in the form of "number of standard doses" rather than monetary sales figures. This approach ensures that the information is not influenced by fluctuations in drug price hikes or discounts.
Lastly, the data are sourced directly from industry — free from government interventions and manipulation. I possess records level data down to stock-keeping units (SKUs) and through various distribution channels, including pharmacies, hospitals, and clinics. I am open to sharing this data with other respected medical freedom fighters for the purpose of validation. However, for the moment, to circumvent potential legal issues such as those Barry Young faced in New Zealand, I have removed the Y-Axis detailing the exact number of doses. Instead, I will be displaying the data as percentages.
Gonadotropin-Releasing Hormone (GnRH) agonists -Zoladex / Lucrin
Let’s start with some old cancer drugs.
Lucrin (leuprolide) and Zoladex (goserelin) belong to the same class of drugs known as Gonadotropin-Releasing Hormone (GnRH) agonists. These drugs work by initially stimulating the release of hormones that control the production of sex hormones (testosterone in men and estrogen in women) from the pituitary gland. After continuous administration, they downregulate the pituitary gland, which leads to a reduction in the sex hormone production.
Both Zoladex, which was approved in 1989, and Lucrin (also known as Lupron in some countries), originating from the 1980s, are very old drugs with a long history of use.
They are supposed to be moving in single digits. However, as illustrated in the data below, their usage trends were consistently in single-digit growth until a sudden upward shift of 13%-15% in 2022.
Chemo Drugs - Docetaxel, Abraxane, Oxaliplatin
Now let’s move on some old chemo drugs, the most common medications for cancers.
The chemo drug Docetaxel, first approved in 1996 for the treatment of various types of cancers, is considered a well-established medication. In terms of its drug lifecycle, the patent for Docetaxel expired in 2010. Typically, these drugs enter a declining stage as doctors opt to prescribe newer generation drugs. However, patients may sometimes choose older drugs based on different factors, such as cost, because generics are available on the market. Take a look at the 48.6% spike in 2022.
Abraxane, known generically as nab-paclitaxel, is another chemotherapy drug that serves as an alternative to Docetaxel and is part of the same family of drugs known as taxanes—indicative from their names ending in "-taxel." Both Docetaxel and nab-paclitaxel work in similar ways but with some distinctions. Approved in 2005, about a decade after Docetaxel, Abraxane represents a more recent iteration within the taxane class.
Abraxane remains a patented medication until 2024, and it has shown a unique trend in its market growth. While most patented drugs nearing the end of their patent don't typically see significant sales increases—since pharmaceutical companies tend to invest marketing dollars on newer drugs with longer remaining patent life—Abraxane defied this norm. In 2019 and 2020, it experienced modest single-digit growth, but in 2022, suddenly, its sales surged by an unexpected 15.7%.
Oxaliplatin, a chemotherapy agent used mainly for colorectal cancer and now off-patent, has seen a significant uptick in use beginning in 2021. Intriguingly, the surge started in that year. It raises the question of whether the initial and subsequent doses of mRNA vaccines could have had an immediate impact on colorectal health.
Interestingly, about five months ago, when I was chatting with Professor Gabriel Oon, the Founding President of the Singapore Society of Oncology, colorectal cancer was pretty much at the top of his mind as seen below.
Brief Intermission
Dear Readers ,
The quest for truth is a challenging one. I launched this Substack in January 2023, and it is now celebrating its first anniversary. While I've made all my content freely available, maintaining this level of output has become increasingly difficult. I've authored over 100 free articles in a year, with the aim of raising awareness and educating the public. I am not affiliated with any 'alternative' media outlets, nor am I affiliated with any other alternative medical organizations; my independence is 100%.
As a former senior executive in Big Pharma, I left the industry in 2020 to caution my friends and family against participating in the mRNA experiments, but three years of unpaid work is beginning to impact me financially. You can discover more about my journey here.
To access the rest of this article, consider signing up for zero obligation 7-day free trial!
Update - I’ve decided to make this article free. But I appreciate any support!
For “Founding Members”, you’ll get my direct telegram for personalized Q&A on any topics including Australian spiders and dropbears.
Continuation…
Here's a brief screenshot of my conversation with Prof Gabriel from August 2023. At that time, I inquired about his expectations concerning particular increases in cancer incidences.
Talking about Colorectal cancer, let’s look at Vectibix known generically as panitumumab, is a monoclonal antibody used as a targeted therapy to treat metastatic colorectal cancer.
Vectibix is often prescribed for patients whose cancer has not responded to or has progressed following other chemotherapy treatments.
Consider the surge of over 30% in 2022 and 2023, which is particularly notable because there wasn't even a decline in 2020 and 2021. For a drug that's 18 years old and approaching the end of its lifecycle, this spike is quite anomalous!
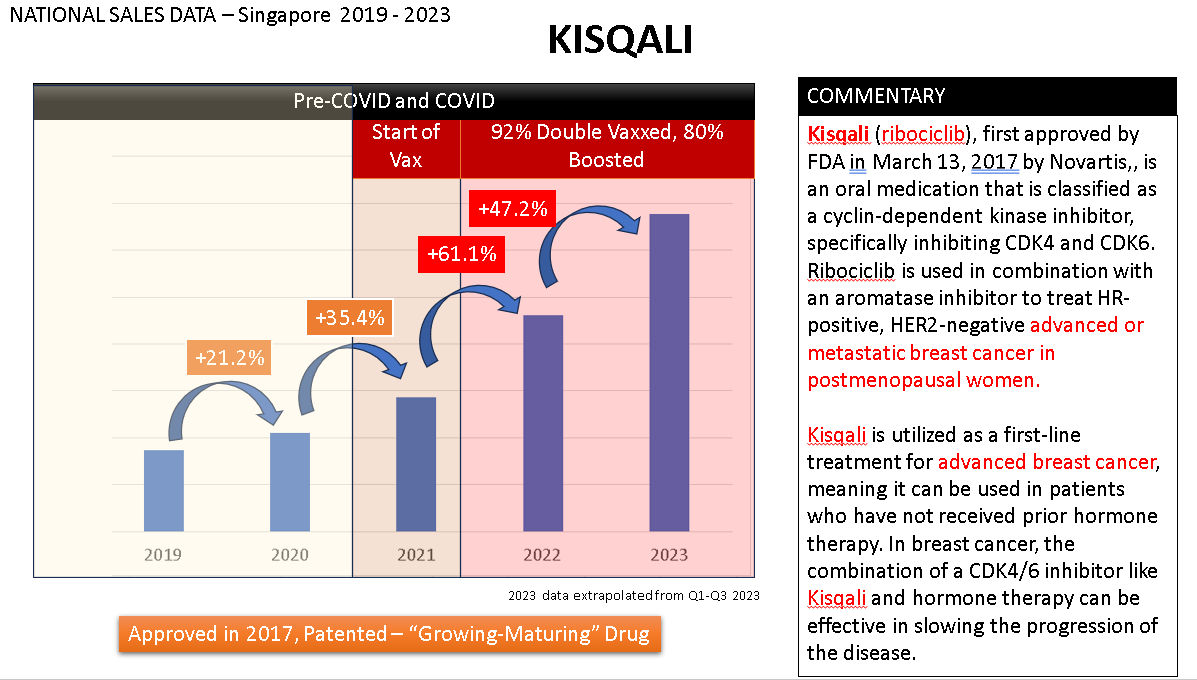
Breast Cancer - Kisqali and Ibrance
Now, let's turn our attention to newer cancer medications. Keep in mind that with these drugs, we are at the initial growth phase of the S-Curve in their lifecycle, so an upward trend in their use is to be anticipated. This uptick can make it more challenging to discern if the rise in cancer cases is related to genetic vaccines. However, by examining the data, you can make your own assessment.
Kisqali is utilized as a first-line treatment for advanced breast cancer. "First-line treatment" simply means it's the initial recommended therapy. If these drugs don’t work, patients then proceed to “second-line” treatments, “third-line”, and so on. Generally, with each increasing “line,” the drugs are "stronger" and come with more side effects. Treatments that are "fourth-line" or beyond are sometimes referred to as "rescue" treatments or "salvage" therapies because earlier options (first-line, second-line, and third-line) have not been effective or have stopped working, and the condition is severe and potentially life-threatening. They are considered a last resort effort because they come with very significant side effects.
As you can see, Kisqali, being a "newer" drug approved in 2017, is still experiencing double-digit growth, although there appears to be a somewhat higher spike of 61% in 2022.
IBRANCE
Ibrance is another medication employed in the treatment of advanced breast cancer, having received approval just two years prior to Kisqali, in 2015. Observing its sales trend from 2019 to 2020, you'll notice that growth is shifting toward single digits. This suggests that Ibrance is nearing its "peak sales" period.
However, it suddenly jumped to 26% in 2022. Again this is a somewhat unusual trend in sales considering there have been no new indications for Ibrance in these years (as sometimes new indications can cause a sudden spike in sales).
Oral Chemo Drug for Brain cancer - Temodal
Next, let’s look at Temodal (generic name temozolomide). This drug is widely recognized as the most frequently used medication for treating brain cancers.
It is a very old drug, developed and approved in 1990s. This drug is quickly and almost completely absorbed from the gut and easily crosses the blood–brain barrier. Therefore, it is very useful for the treatment of different types of brain cancers.
Do you know know what else crossed the blood-brain barrier? Yes, these Lipid Nanoparticles encapsulated mRNAs (and we now know it has some bonus endotoxins i.e DNA plasmid contaminations!)
Anyway, there was a significant decline in usage during 2020, the first year of COVID-19, possibly as a result of reduced access to treatment amidst the lockdowns. However, there was a dramatic increase doses administered in 2020 to 2022, an increase of more than 3x! This is a 20+ year old drug and it shouldn’t go up by 300% in a span of two years just because of some missed diagnosis in a single year.
Blood & Bone Cancer - Tasigna, Kyprolis, Jakafi, Imbruvica
Tasigna is an established drug used to treat a type of blood cancer known as Chronic Myeloid Leukemia. Approved in 2007, it is still protected by patent, but it supposedly reached peak sales around 2019/2020, as indicated by the single-digit sales growth. However, in 2022, its sales unexpectedly jumped by 16.9%.
Jakavi, known as Jakafi in the US, is prescribed for certain blood or bone marrow cancers. Once more, there was a significant and sudden increase of 30% in its usage in 2022.
Kyprolis, which is a slightly newer drug compared to Jakafi, is administered for the treatment of multiple myeloma (blood cancer). In 2022, it too experienced a substantial increase, with a spike of 49.6%.
Imbruvica, which is now under the ownership of Johnson & Johnson(Janssen Pharmaceuticals) and is utilized in the treatment of several types of blood cancers, also saw an uptick in use with a sudden 17.2% increase in 2022.
Kidney Cancer - Inlyta
Next, let’s look at Inlyta that is used for Kidney cancer. It is a drug that is approved in 2012, but you can see that they had a huge growth in 2020. This is because in 2019, it received a new indication when FDA approved it as a first line treatment (historically it was second line), so it has a huge sales jump of 83% in 2019. It then stabilized in 2021. The interesting part is that the sales suddenly doubled in 2023.
Note that the growth rates of various cancer drugs differ; some show increases in 2021, others in 2022, and yet others in 2023. I am sure there are explanations, which I will look into at a later stage.
Warnings from Oncologists and cancer experts.
Below you'll find notable U-Turns and words of caution from distinguished oncologists and cancer experts. Click on their names to explore in detail.
Prof Angus Dalgleish, Professor of Oncology at St Georges Hospital Medical School London / Foundation chair of oncology at St George's, University of London.
Prof Gabriel Oon, Founding President of Singapore’s Oncology Society and former WHO consultant.
Prof Wafik El-Deiry, Director of the Cancer Center at Brown University, Associate Dean for Oncologic Sciences at the Warren Alpert Medical School.
Prof Phillip Buckhaults, Professor of cancer genomics from University of South Carolina.
Other data points - Professor Konstantine Beck’s presentation on Swiss data that individuals on cancer medication has doubled in 2022.
Professor Konstantine Beck of the University of Lucerne recently highlighted a striking fact: the number of individuals on cancer medication has surged, with a 94% increase reported in 2022, effectively doubling from previous years.
University of Lucerne's Prof. Dr. Konstantin Beck Raises Alarm Over Surge in Swiss Cancer Cases in 2022
Switzerland, reputed for its high standard of living and healthcare system, encountered an alarming upsurge in cancer cases in 2022. In a presentation (translated video above) by health economist Prof Dr Konstantin Beck of the University of Lucerne on 5th Dec 2023, there was a striking increase magnitudes higher than population growth factors could expl…
Dr Joseph Ladapo calling for a halt on mRNA vaccines citing DNA contaminations and cancer risks.
Dr Joseph Ladapo, Surgeon General of Florida recently (January 3rd 2024) recently called for the halt of mRNA vaccines citing DNA contaminations.
Full statement available here.
Here are some the highlights:
The Surgeon General outlined concerns regarding nucleic acid contaminants in the approved Pfizer and Moderna COVID-19 mRNA vaccines, particularly in the presence of lipid nanoparticle complexes, and Simian Virus 40 (SV40) promoter/enhancer DNA. Lipid nanoparticles are an efficient vehicle for delivery of the mRNA in the COVID-19 vaccines into human cells and may therefore be an equally efficient vehicle for delivering contaminant DNA into human cells. The presence of SV40 promoter/enhancer DNA may also pose a unique and heightened risk of DNA integration into human cells.
DNA integration could theoretically impact a human’s oncogenes – the genes which can transform a healthy cell into a cancerous cell.
DNA integration may result in chromosomal instability.
The Guidance for Industry discusses biodistribution of DNA vaccines and how such integration could affect unintended parts of the body including blood, heart, brain, liver, kidney, bone marrow, ovaries/testes, lung, draining lymph nodes, spleen, the site of administration and subcutis at injection site.
Parting Thoughts…
In all my life in Pharma, I have sometimes seen sales anomalies in certain drug classes. For instance, when the painkiller Vioxx (rofecoxib) was recalled on September 30, 2004, other drugs in the "coxib" class, like Celebrex (celecoxib) and Bextra (valdecoxib), suffered a drop in sales because the market became more cautious with COX-2 inhibitors (coxib). It's also not uncommon to see entire drug classes increase in sales, like statins in the 2000s when Pharma invested a lot of money to grow the entire statin drug class (led by Pfizer's atorvastatin, or Lipitor).
However, I have never seen such a spike occur within a single year (2022), for a single disease (cancer) across multiple drug classes, both new and old drugs, and across various companies. This is striking and adds to a long list of data points, suggesting that the experimental mRNAs are causing great harm. Slowly but surely, we are also starting to understand the mechanisms (DNA contamination, SV40 promoters, stem cell injuries, immunosuppression, etc.).
And get this: I am only looking at cancer drugs; I haven't even done a deep dive into cardiac and neuro drugs yet.
I trust that you'll gain valuable insights from this exclusive Aussie17 report!
Should you find it beneficial, I would appreciate it if you consider purchasing a gift subscription for someone else who might learn from this information. Your support would also be a great help to me!
Signing off for now
A17






















Personally I can see that this article had HOURS of time put into it...and, to me, that is a welcome change to most channels and their content on substack which seems to be generated quickly and with little attention to detail. So, well done. And please keep this cancer drug numbers coming. It is evidence of the greatest crime ever committed.
Brilliant work! Very disturbing. Unfortunately totally expected! Shared!