Part 2 of Shit Hitting The Fan!
and Kevin Mckernan's makes it official with the FDA!
Update: Ops, I made a mistake with the number of doses administered! Fixed it! Time to take a break!
[Read Part 1 here]
It's great to see this news gaining more traction,it is crucial. While many of us have been focused on VAERS over the past two years, it is even more important to shed light on Pfizer's 393 pages of Adverse Events. Allow me to explain why through a Q&A format.
Before we proceed, I want to mention a caveat: I am not a Pharmacovigilance(“PV”) expert. Therefore, most of what I'm about to share is based on my interactions with former PV colleagues.
Is this VAERS Data?
No, this is not VAERS.
So, what is the difference between this and VAERS?
VAERS is a U.S.-based system used for capturing Vaccine Adverse Events. It allows individuals, healthcare providers, and vaccine manufacturers to report any adverse events that occur following vaccination. However, it is important to note that VAERS is not a verified system.
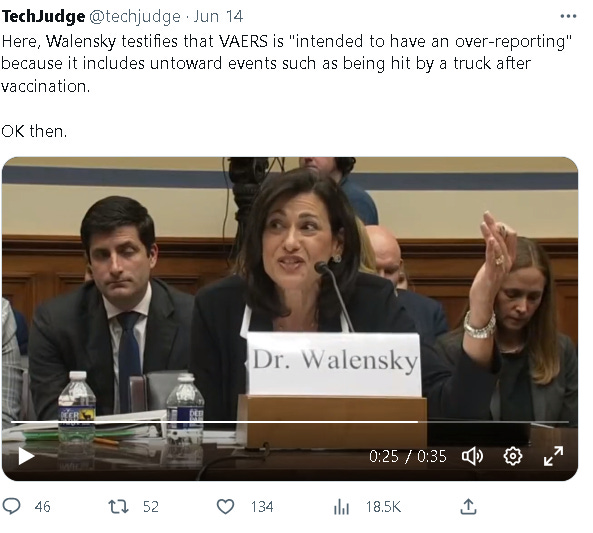
Due to its unverified nature, CDC Director Rochelle Walensky has stated that VAERS may have an over-reporting of adverse events. She mentioned that examples like "people getting hit by a truck" are included in VAERS, highlighting the range of reports it collects.
On the other hand, pharmaceutical companies employ robust pharmacovigilance systems to capture adverse events associated with their products. These systems involve a more comprehensive and thorough process of data collection, assessment, and verification.
In essence, internal Adverse Event data is verified. This means that whenever they receive a report, there is someone internally verifying the accuracy of the data. In some cases, they may reach out to the patient or doctor to gather additional information (for example, if the batch number was omitted or if the Adverse Event was not described clearly).
Is this 393-page Adverse Event underreported?
In my opinion, yes. It shares similarities with the underreporting issues seen in VAERS. However, there are also systematic methods employed by Big Pharma to deliberately underreport Adverse Events. I have discussed some of these in Pharmafiles #11, which you can find here.
Is the 393-page Adverse Event document confidential? Was it leaked?
There is some nuance to the answer. Allow me to explain in simpler terms.
In highly regulated industries like the pharmaceutical sector, document management follows a meticulous and labor-intensive process. Unlike simply opening Microsoft Word and typing away, creating a new document in Pharma involves setting various metadata associated with it.
One important aspect is the document classification, which may include options like
[ ] Public
[ ] Restricted
[ ] Private
[ ] Confidential
[ ] Secret.
Each classification has specific rules attached to it. "Public" documents, such as Annual Reports, are accessible to everyone. "Restricted" documents may be limited to employees and contractors/suppliers. "Private" documents may be restricted to employees only. "Confidential" documents may be available to custom-defined groups, such as regulators. The release of these documents to the public is typically determined by regulators, and acquiring them may involve additional processes like Freedom of Information (FOI) requests. It's important to note that "Confidential" does not necessarily mean leaked; it often means the document is subject to certain restrictions.
The documentation systems in place are complex, they give me nightmares up to this day. "Documentum" is commonly used in the big pharma I worked with, and tech geeks may have more insights about it.
Anyway, thanks to Twitter, I recently became aware that an earlier version of this document (based on April 2022 updates) was available. Surprisingly, the news didn't cover it extensively, and Senator Rennick suggested that it was heavily censored. However, Senator Rennick obtained it through a Freedom of Information request. You can find the link to the tweet below.
I was also informed that the breaking of the 393 Pages initially came from LawyerLisa here.
What are Spontaneous vs Non-Spontaneous AEs?
Spontaneous adverse events occur when individuals independently report negative reactions to drugs. On the other hand, non-spontaneous adverse events are identified through meticulous studies, such as clinical trials, or through monitoring conducted by researchers or drug companies.
Regarding the 5 million adverse events, how many doses of Pfizer's mRNA vaccine were administered in relation to these cases?
2,693,922,584 administered doses. [Source]
To put this into perspective, there have been 4,964,106 adverse events reported, indicating a 0.184% chance of an adverse event for every administered dose. This means approximately 1 adverse event occurs for every 543 doses administered!
Now, using Senator Rennick's numbers from the April 2022 update, I calculated the ratio of Serious AEs to Non-serious AEs.
With 28.8% serious AEs, it appears that we are observing 1 Serious Adverse Event for every 1887 doses administered.
This is above Fraiman’s paper (1 in 800 serious adverse events), maybe it could be explained by the underreporting.
1 in 2000 doses for every SAE is still a very bad ratio!
Perhaps someone else can verify these figures. Let's pass the baton to Jessica Rose, who meticulously tabulated the data from the 393 pages and organized it in an Excel spreadsheet!
In other news…
Kevin McKernan recently presented evidence of DNA contamination to the FDA. You can watch the video below for more details.
I believe this is a significant issue, as it involves an Out of Spec situation. Both the FDA and Pfizer are aware of it, and now it has been officially documented. Multiple laboratories across the globe have replicated Kevin's findings, which further validates the concerns. An immediate investigation followed by a product recall is necessary.
You can read more about it by following this link.
Signing off for now!!
-A17
PS: Please share my post or subscribe, all my posts are free! Or just drop a comment! It will give me some motivation to write!








Thank you Aussie17 for all you do. I am a big fan of yours.
In claiming VAERS is over-reported and that a traffic accident after vaccination could be reported as vax injury/death, Walenski is confusing, somewhat disingenuously, VAERS deaths/injuries with alleged covid deaths which we know were ridiculously and fraudulently inflated exactly in this way. How could that senator have let her get away with this? If it had been me questioning her, I would have wiped the smirk off her face.